Buffer In Human Blood . human blood has a buffering system to minimize extreme changes in ph. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. It consists of a solution of a weak acid and its. A buffer is a system that resists a change in ph. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. In this system, gaseous metabolic. a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. One buffer in blood is based on the presence.
from animalia-life.club
A buffer is a system that resists a change in ph. It consists of a solution of a weak acid and its. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. One buffer in blood is based on the presence. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. human blood has a buffering system to minimize extreme changes in ph. In this system, gaseous metabolic.
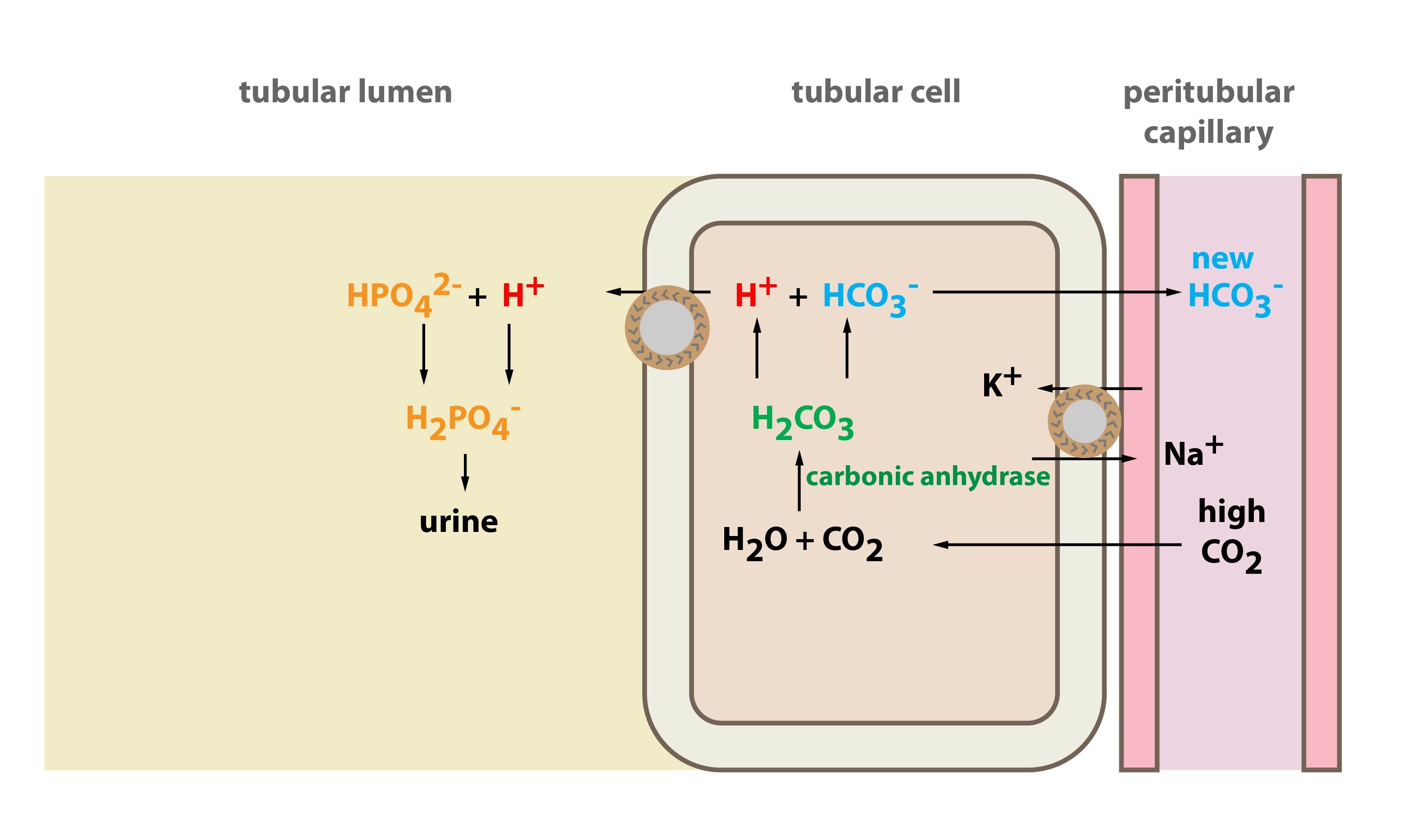
Phosphate Buffer System Equation
Buffer In Human Blood a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. It consists of a solution of a weak acid and its. a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. A buffer is a system that resists a change in ph. One buffer in blood is based on the presence. human blood has a buffering system to minimize extreme changes in ph. In this system, gaseous metabolic.
From www.slideshare.net
02 hydrolysis. buffers__colloids Buffer In Human Blood normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. A buffer is a system that resists a change in ph. It consists of a solution of a weak acid and its. One buffer in blood is based on the presence. In this system, gaseous metabolic. human blood has a. Buffer In Human Blood.
From www.slideserve.com
PPT Buffers and Titrations PowerPoint Presentation, free download Buffer In Human Blood A buffer is a system that resists a change in ph. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. It consists of a solution of a weak acid. Buffer In Human Blood.
From facts.net
20 Fascinating Facts About Blood Buffer Buffer In Human Blood One buffer in blood is based on the presence. A buffer is a system that resists a change in ph. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. human blood has a buffering system to minimize extreme changes in ph. a variety of buffering systems permits blood. Buffer In Human Blood.
From www.slideserve.com
PPT Buffers in Blood. Acidosis and Alkalosis. PowerPoint Presentation Buffer In Human Blood One buffer in blood is based on the presence. In this system, gaseous metabolic. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. a variety of buffering systems permits blood and. Buffer In Human Blood.
From animalia-life.club
Phosphate Buffer System Equation Buffer In Human Blood A buffer is a system that resists a change in ph. It consists of a solution of a weak acid and its. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. In this system, gaseous metabolic. a variety of buffering systems permits blood and other bodily fluids to maintain a. Buffer In Human Blood.
From www.chegg.com
Solved Human blood contains several buffering systems, the Buffer In Human Blood a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. One buffer in blood is based on the presence. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. It consists of a solution of a weak acid and. Buffer In Human Blood.
From www.slideserve.com
PPT Buffers in Blood. Acidosis and Alkalosis. PowerPoint Presentation Buffer In Human Blood the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. human blood has a buffering system to minimize extreme changes in ph. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. A buffer is a system that resists a change in. Buffer In Human Blood.
From www.pinterest.com
Physiology Blood Buffer System Behrouz Human body facts Buffer In Human Blood the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. In. Buffer In Human Blood.
From www.slideserve.com
PPT Physiological system of blood. Functional importance of blood Buffer In Human Blood human blood has a buffering system to minimize extreme changes in ph. It consists of a solution of a weak acid and its. a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. A buffer is a system that resists a change in ph. One buffer in. Buffer In Human Blood.
From www.slideserve.com
PPT Biological Buffers PowerPoint Presentation, free download ID Buffer In Human Blood a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. One buffer in blood is based on the presence. It consists of a solution of a weak acid and. Buffer In Human Blood.
From www.slideserve.com
PPT General, Organic, and Biochemistry, 7e PowerPoint Presentation Buffer In Human Blood a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. human blood has a buffering system to minimize extreme changes in ph. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. In this system, gaseous metabolic. A buffer. Buffer In Human Blood.
From www.ehow.com
What Are the Three Buffer Systems in Body Fluid? Healthfully Buffer In Human Blood a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. A buffer is a system that resists a change in ph. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. human blood has a buffering system to minimize. Buffer In Human Blood.
From www.slideserve.com
PPT Buffers in Blood. Acidosis and Alkalosis. PowerPoint Presentation Buffer In Human Blood a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. It consists of a solution of a weak acid and its. A buffer is a system that resists a change in ph. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic. Buffer In Human Blood.
From www.slideserve.com
PPT RESPIRATORY SYSTEM 3 PowerPoint Presentation, free download Buffer In Human Blood the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. human blood has a buffering system to minimize extreme changes in ph. a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. It consists of a solution of a. Buffer In Human Blood.
From www.youtube.com
Buffer action in the blood YouTube Buffer In Human Blood a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. A buffer is a system that resists a change in ph. One buffer in blood is based on the. Buffer In Human Blood.
From www.slideserve.com
PPT Buffer Systems of the Body PowerPoint Presentation, free download Buffer In Human Blood A buffer is a system that resists a change in ph. In this system, gaseous metabolic. human blood has a buffering system to minimize extreme changes in ph. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without being pathological. One buffer in blood is based on the presence. a variety. Buffer In Human Blood.
From slideplayer.com
Should we control a chemical that ppt download Buffer In Human Blood In this system, gaseous metabolic. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. a variety of buffering systems permits blood and other bodily fluids to maintain a narrow ph range, even in the face. It consists of a solution of a weak acid and its. normal blood ph. Buffer In Human Blood.
From www.youtube.com
Buffer in the Human Blood? What is Buffer in Chemistry? YouTube Buffer In Human Blood It consists of a solution of a weak acid and its. One buffer in blood is based on the presence. the buffer systems functioning in blood plasma include plasma proteins, phosphate, and bicarbonate and carbonic acid buffers. In this system, gaseous metabolic. normal blood ph is 7.4, and arterial ph may only vary between 7.35 and 7.45 without. Buffer In Human Blood.